High-sensitivity Troponin-T: Difference between revisions
mNo edit summary |
mNo edit summary |
||
| Line 1: | Line 1: | ||
As of November 12, 2019, the cardiac troponin assay utilized by the Yukon-Kuskokwim Delta Regional Hospital (YKDHR) is the Roche Diagnostics [https://diagnostics.roche.com/us/en/products/params/elecsys-troponin-t-high-sensitive-tnt-hs.html ''Elecsys® Troponin T Gen 5 STAT'']. This is a high-sensitivity fifth-generation cardiac troponin assay. | As of November 12, 2019, the cardiac troponin assay utilized by the Yukon-Kuskokwim Delta Regional Hospital (YKDHR) is the Roche Diagnostics [https://diagnostics.roche.com/us/en/products/params/elecsys-troponin-t-high-sensitive-tnt-hs.html ''Elecsys® Troponin T Gen 5 STAT'']. This is a high-sensitivity fifth-generation cardiac troponin assay. | ||
Cardiac troponin assays measure the concentration of either troponin-T or troponin-I. Both high-sensitivity and | Cardiac troponin assays measure the concentration of either troponin-T or troponin-I. Both high-sensitivity and traditional troponin assays measure the exact same molecule, but high-sensitivity assays measure ''much'' more precisely and at ''much'' lower concentrations. The high-sensitivity and traditional assays can be distinguished by the units in which they are reported: high-sensitivity assays are reported in ng/'''L''' whereas the traditional assays are reported in ng/'''mL'''. | ||
:::Conversion between the results can be done by moving the decimal point three places: a | :::Conversion between the results can be done by moving the decimal point three places: a traditional concentration of 0.4 ng/mL is equivalent to a high-sensitivity concentration of 400 mg/mL, and a high-sensitivity concentration of 14 ng/L is equivalent to a traditional concentration of 0.014 ng/L. This latter conversion illustrates that the high-sensitivity assays accurately measure concentrations which are ''two orders of magnitude'' lower than the assay we were previously using. | ||
Though high-sensitivity troponin assays have been used in Europe and Canada since approximately 2009, the first U.S. FDA approval occurred in 2017. | Though high-sensitivity troponin assays have been used in Europe and Canada since approximately 2009, the first U.S. FDA approval occurred in 2017. | ||
| Line 16: | Line 16: | ||
<br> | <br> | ||
<br> | <br> | ||
== Level versus Delta ( | == Level versus Delta (?) == | ||
With | With traditional troponin assays, a troponin level was not detectable in most patients who lack cardiac ischemia. Therefore the ''level'' of the troponin was the focus while the subsequent ''delta (?)'' (i.e. a change) was a secondary consideration. | ||
Due to the high-sensitivity assays' ability to detect a troponin level in most patients who lack cardiac ischemia, many experts recommend a reversal in diagnostic thinking: for most patients the delta ( | Due to the high-sensitivity assays' ability to detect a troponin level in most patients who lack cardiac ischemia, many experts recommend a reversal in diagnostic thinking: for most patients the delta (?) should be the focus while the level is a secondary consideration.<ref>Helman, A. McRae, A. Lang, E. Low Risk Chest Pain and High Sensitivity Troponin – A Paradigm Shift. Emergency Medicine Cases. July, 2019. https://emergencymedicinecases.com/low-risk-chest-pain-high-sensitivity-troponin. Accessed December 2, 2019.</ref> | ||
However, there are initial levels below and above which a delta is not required: | However, there are initial levels below and above which a delta is not required: | ||
| Line 26: | Line 26: | ||
<br> | <br> | ||
<br> | <br> | ||
== Relative versus Absolute Delta ( | == Troponin Delta == | ||
With | === Relative versus Absolute Delta (?) === | ||
With traditional troponin assays, a delta of 20% was often used to rule-in ACS. | |||
However, common situations arise with high-sensitivity troponin assays where relative deltas can lead to both over- and underdiagnosis. For example, if the initial troponin level is 5 ng/L, then a delta of 1 ng/L is a 20% rise, despite the change being within the expected margin of error of the assay. Alternately, the 20% relative change can miss ACS in patients with chronically elevated troponin levels. | However, common situations arise with high-sensitivity troponin assays where relative deltas can lead to both over- and underdiagnosis. For example, if the initial troponin level is 5 ng/L, then a delta of 1 ng/L is a 20% rise, despite the change being within the expected margin of error of the assay. Alternately, the 20% relative change can miss ACS in patients with chronically elevated troponin levels. | ||
| Line 34: | Line 35: | ||
<br> | <br> | ||
<br> | <br> | ||
== | <br> | ||
=== Delta Cutoffs === | |||
The YKDHR Laboratory recommends that a ?1h > 3 ng/L be used to rule-in ACS. | |||
<br> | <br> | ||
<br> | <br> | ||
== Cutoffs == | |||
== Troponin Level == | |||
=== Level Cutoffs === | |||
Hs-cTnT is considered "positive" when above the gender-specific 99th percentile URL (upper reference range). Per eMail from Scott Cox (YKDHR Director of Diagnostic Services) on 11/10/2019, the following cutoff are recommended for our assay: | Hs-cTnT is considered "positive" when above the gender-specific 99th percentile URL (upper reference range). Per eMail from Scott Cox (YKDHR Director of Diagnostic Services) on 11/10/2019, the following cutoff are recommended for our assay: | ||
{| class="wikitable" style="margin: auto;" | {| class="wikitable" style="margin: auto;" | ||
| Line 52: | Line 52: | ||
|} | |} | ||
<br> | <br> | ||
?1h >= '''3''' ng/L is considered positive for acute myocardial injury (AMI).<br> | |||
<br> | <br> | ||
Per the ACC white paper (see below):<br> | Per the ACC white paper (see below):<br> | ||
Revision as of 18:27, 3 December 2019
As of November 12, 2019, the cardiac troponin assay utilized by the Yukon-Kuskokwim Delta Regional Hospital (YKDHR) is the Roche Diagnostics Elecsys® Troponin T Gen 5 STAT. This is a high-sensitivity fifth-generation cardiac troponin assay.
Cardiac troponin assays measure the concentration of either troponin-T or troponin-I. Both high-sensitivity and traditional troponin assays measure the exact same molecule, but high-sensitivity assays measure much more precisely and at much lower concentrations. The high-sensitivity and traditional assays can be distinguished by the units in which they are reported: high-sensitivity assays are reported in ng/L whereas the traditional assays are reported in ng/mL.
- Conversion between the results can be done by moving the decimal point three places: a traditional concentration of 0.4 ng/mL is equivalent to a high-sensitivity concentration of 400 mg/mL, and a high-sensitivity concentration of 14 ng/L is equivalent to a traditional concentration of 0.014 ng/L. This latter conversion illustrates that the high-sensitivity assays accurately measure concentrations which are two orders of magnitude lower than the assay we were previously using.
Though high-sensitivity troponin assays have been used in Europe and Canada since approximately 2009, the first U.S. FDA approval occurred in 2017.
NOTE: The information below is not a guideline, but rather excerpts and links intended to augment and/or help develop clinical judgement.
Definition
To qualify as high-sensitivity (or highly-sensitive), a cardiac troponin assay must be able to detect a level of cardiac troponin in >50% of normal individuals.[1] This means that the majority of patients without cardiac ischemia will have a detectable level of cardiac troponin. This has substantial implications for how clinician conceptualize and interpret cardiac troponin levels.
Level versus Delta (?)
With traditional troponin assays, a troponin level was not detectable in most patients who lack cardiac ischemia. Therefore the level of the troponin was the focus while the subsequent delta (?) (i.e. a change) was a secondary consideration.
Due to the high-sensitivity assays' ability to detect a troponin level in most patients who lack cardiac ischemia, many experts recommend a reversal in diagnostic thinking: for most patients the delta (?) should be the focus while the level is a secondary consideration.[2]
However, there are initial levels below and above which a delta is not required:
- If >= 3 hours since onset of symptoms and a normal EKG, then an initial troponin level < 6 ng/L can rule-out ACS.
- In the context of new symptoms consistent with ACS, an initial troponin level >100 ng/L rules-in ACS.
Troponin Delta
Relative versus Absolute Delta (?)
With traditional troponin assays, a delta of 20% was often used to rule-in ACS.
However, common situations arise with high-sensitivity troponin assays where relative deltas can lead to both over- and underdiagnosis. For example, if the initial troponin level is 5 ng/L, then a delta of 1 ng/L is a 20% rise, despite the change being within the expected margin of error of the assay. Alternately, the 20% relative change can miss ACS in patients with chronically elevated troponin levels.
Therefore many experts recommend using an absolute delta to rule-out or rule-in ACS.
Delta Cutoffs
The YKDHR Laboratory recommends that a ?1h > 3 ng/L be used to rule-in ACS.
Troponin Level
Level Cutoffs
Hs-cTnT is considered "positive" when above the gender-specific 99th percentile URL (upper reference range). Per eMail from Scott Cox (YKDHR Director of Diagnostic Services) on 11/10/2019, the following cutoff are recommended for our assay:
| Women | >= 14 ng/L |
| Men | >= 22 ng/L |
?1h >= 3 ng/L is considered positive for acute myocardial injury (AMI).
Per the ACC white paper (see below):
- A single Hs-cTnT >= 100 ng/L is diagnostic of AMI (in the appropriate clinical context).
- When chest pain has been present for >= 2 hours, a single Hs-cTnT < 6 ng/L has been reported to rule out AMI with essentially 100% negative predictive value.
Diagnostic Algorithms
The following diagnostic algorithm has been proposed for high-sensitivity cardiac troponin-T by the ACC (American College of Cardiology), though they caution that "The safety of this approach is currently unclear":[1]
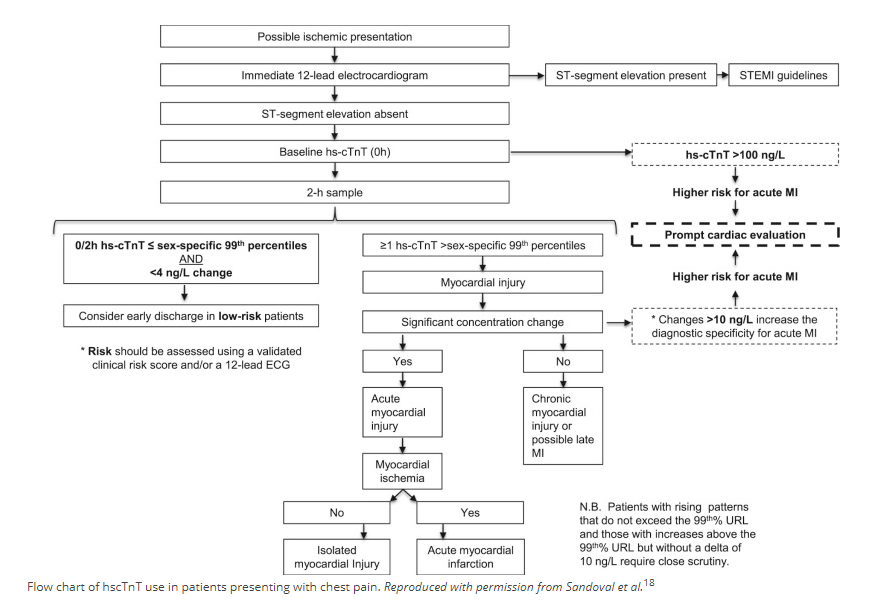
The internet contains several somewhat similar algorithms on reputable medical sites. In particular, the ACEP Webinar (see below, page 16) displays the following diagnostic algorithm:
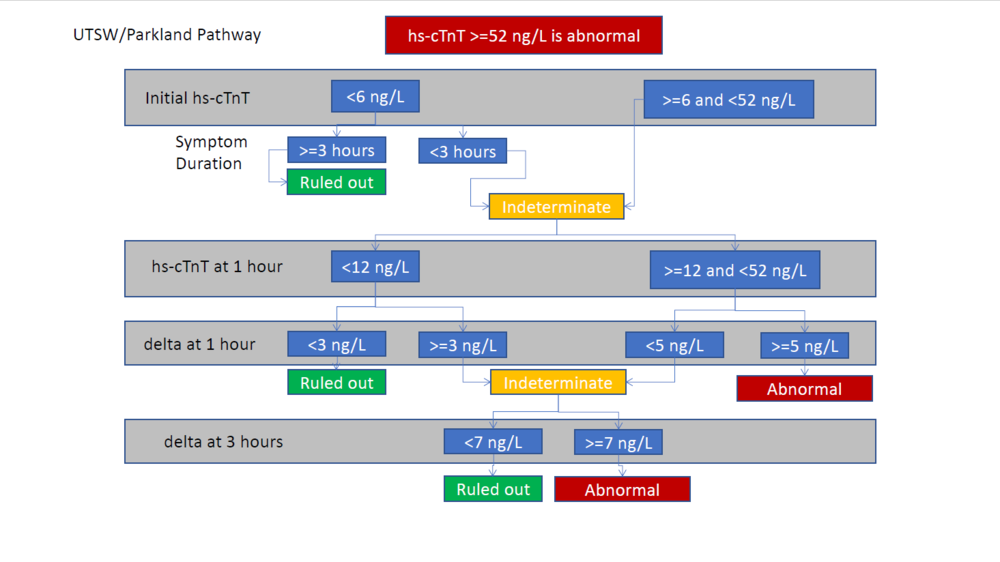
Note that this algorithm does not include risk stratification, such as with the HEART score. Yet the same Webinar contains other algorithms which separate out Hs-cTnT and call the risk stratification a HEAR score (or "modified-HEART" score) and guide diagnosis/management based upon different combinations of Hs-cTnT and HEAR values. Yet other algorithms in the same Webinar recommend skipping risk stratification in those who have low- or high-risk EKG/Hs-cTnT results and only risk stratifying those with intermediate-risk EKG/Hs-cTnT results.
Helpful Links
ACC (American College of Cardiology)
- High-Sensitivity Cardiac Troponin in the Evaluation of Possible AMI (July 16, 2018)
- SUMMARY: Fourth Universal Definition of Myocardial Infarction (Aug 25, 2018)
ACEP
- Webinar: Incorporating High-Sensitivity Troponin into Your ED
- Critical Issues in the Evaluation and Management of Emergency Department Patients with Suspected Non–ST-Elevation Acute Coronary Syndromes (Jun 2018)
MD Calc
References
- ↑ 1.0 1.1 High-Sensitivity Cardiac Troponin in the Evaluation of Possible AMI. American College of Cardiology. https://www.acc.org/latest-in-cardiology/articles/2018/07/16/09/17/high-sensitivity-cardiac-troponin-in-the-evaluation-of-possible-ami. Accessed December 3, 2019.
- ↑ Helman, A. McRae, A. Lang, E. Low Risk Chest Pain and High Sensitivity Troponin – A Paradigm Shift. Emergency Medicine Cases. July, 2019. https://emergencymedicinecases.com/low-risk-chest-pain-high-sensitivity-troponin. Accessed December 2, 2019.