Cardiac Biomarkers in Pediatrics
Bottom Line Up Front (BLUF):
- Compared to healthy adults, the serum troponin and BNP levels in healthy neonates and infants less than 1 year have normal ranges with 99th percentiles nearly two orders of magnitude greater.
- HIGH-SENSITIVITY TROPONIN-T (hs-cTnT)
- As a biomarker, troponin occupies a relatively unique position in medicine. Unlike most biomarker cut-offs, which are based upon a desired sensitivity/specificity for diagnosis of a condition, troponin cut-offs are an arbitrarily chosen percentile of the values observed in a healthy population. A troponin value above the 99th percentile in healthy persons is the DEFINITION of "myocardial injury" (regardless of the clinical situation of the individual patient). Therefore, the troponin cut-off does not have a sensitivity/specificity for "myocardial injury," it simply IS myocardial injury. This is a relatively unique role for a serum biomarker, outranking even the concept of "gold standard".
- As of January 2022, studies have only recently begun to quantify the distributions and normal ranges of troponin levels in healthy infants and children. At less than one year of age, the 99th percentile is markedly higher than in adults. At around one year the 99th percentiles are similar, and after one year the 99th percentiles are lower than in adults. Though sex-adjusting the 99th percentiles is not problematic, age-adjusting is: a hs-cTnT of 70 would be POSITIVE at 60 days old but NEGATIVE at 61 days old.[1] In such a setting, decision making is less clear and the definition of "myocardial injury" starts seeming rather arbitrary (if not meaningless).
- Myocardial injury is NOT synonymous with ischemia. Numerous non-ischemic causes of myocardial injury exist, such as infiltrative diseases, trauma, inflammation, etc. In fact, non-ischemic etiologies are greatly predominant in all pediatric age groups.[2]
- N-TERMINAL pro-BNP (N-Pro-BNP): Diagnostic cut-offs have been published for several disease conditions in some pediatric age groups, but few have been validated.
- Indeed, considering the markedly higher normal ranges, and as argued by Assandro et al in 2013,[3] these cardiac biomarkers may lack the ability to distinguish disease from non-disease in some (or all) pediatric age groups.
- No one should order a cardiac biomarker in a pediatric patient with the intent to rule-in or rule-out disease without:
- Evidence that the marker can distinguish disease from non-disease (as represented by the AUROC) in the age group in question.
- Knowledge of the diagnostic cut-off for the disease in question, as well as the sensitivity and specificity associated with that cut-off.
- False-positive interpretation of cardiac biomarkers can lead to unnecessary invasive testing, which is itself a harm, but which also carries a non-negligible risk of additional harm (i.e. procedural complications).
- If a consultant recommends ordering a troponin or BNP in one of these age groups, the consulting provider should inquire about the diagnostic cut-off,† the sensitivity, and the specificity; if the consultant cannot provide this information, further research and/or additional consultants should be strongly considered prior to ordering these tests. There is very little clinical utility to a test result which no one knows how to interpret, and there is a risk of harm from a test result which is likely to be misinterpreted.
- This page is not applicable to the following topics:
- Following cardiac biomarker trends for an already-diagnosed condition.
- Use cardiac biomarkers in preterm infants.
hs-cTnT
Biochemistry
The troponin Wikipedia article provides an excellent, succinct review of the biochemistry of troponin. In summary, the troponin complex is a molecule bound to myosin within muscle cells and involved in muscle contraction. Troponin-I and troponin-T are specific to cardiac muscle.
Troponin "leaks" from cardiomyocytes when cell membrane permeability is increased; this condition is termed myocardial "injury," and it can be caused by both ischemic and non-ischemic processes. Ischemic processes include acute coronary syndrome and chronic coronary ischemia (such as ischemic cardiomyopathy), while non-ischemic processes include a multitude of diverse conditions (such as inflammation, apoptosis, cytotoxicity, infiltrative diseases, hypoxemia, shock, ventricular strain, trauma, etc).[4] Clinical decisions regarding elevated troponin must incorporate an understanding non-ischemic myocardial injury. Indeed, while ischemic causes of myocardial injury predominate in older adults, non-ischemic causes vastly predominate in all pediatric age groups.[2]
Normal Range[s] in Pediatric Age Groups
Only studies reporting values for the Roche hs-cTnT Gen 5 STAT assay are presented here (because other assays and/or troponin types have different cut-offs and are thus not pertinent to clinical decision making at YKDRH). Search methods are documented in the Methods section (below).
- Jehlicka et al (2018)[5]
- Prospective, single-cohort study of cord blood of 241 healthy, term newborns in the Czech Republic. The authors report means and quartile values similar to other studies of hs-cTnT, but they report 97.5th percentiles rather than 99th percentiles. These results help understand the distribution newborn hs-cTnT levels, but without the 99th percentiles use of this data is limited and thus not presented here. However, the authors report several notable findings:
- "The difference between hscTnT concentrations in boys and girls tended to be statistically significant (38.7 ng/l [33.0 to 52.8 ng/l] and 36.7 ng/l [29.2 to 47.1 ng/l], respectively, p=0.052)."
- "The concentration of hscTnT was found to be significantly lower in newborns delivered via Cesarean section when compared with vaginal delivery (35.0 ng/l [28.5 to 43.4 ng/l] and 38.9 ng/l [32.6 to 48.4 ng/l], respectively, p=0.0084)."
- "The hscTnT level correlated inversely with the base status (BE: rho=-0.14, p=0.03) and with the pH level of the umbilical cord blood (rho=-0.14, p=0.03)."
- "Elevated plasma concentrations of hscTnT decreased to adult range within six months."
- Bohn et al (2019)[6]
- Unclear if retrospective or prospective; samples from "apparently" healthy infants (<1y) obtained from local maternity wards and outpatient clinics in Toronto, Canada; samples from >1 obtained from the "CALIPER biobank." Table-1 shows the reported 99th percentiles. Additionally, the authors report several notable observations:
- "hs-cTnT concentrations were markedly increased from 0 to <6 months and subsequently decreased and narrowed at 1 year."
- "Although hs-cTnT concentrations from 0 to <1 year were normally distributed, hs-cTnT concentrations for both sexes of age 1 to <19 years were highly skewed."
- The authors comment about their 1y to 19y age group having 99th percentile cut-offs of 11 (F) and 14 (M) and this differing from the manufacturer package insert. They astutely note that the major cause for this difference is likely their use of a different reference population, and they highlight that neither reference population had imaging done (i.e. structural and coronary cardiac evaluations) to show that they were truly healthy with regard to establishing a distribution in "healthy" individuals.
| Age | n | 99th percentile |
|---|---|---|
| 0 – <6m | 55 | 93 |
| 6m – <1y | 44 | 21 |
| 1 – <19y F | 249 | 11 |
| 1 – <19y M | 249 | 14 |
- Karlen et al (2019)[7]
- Prospective, inpatient, single-cohort study of 158 [expected] healthy, term newborns, in Stockholm, Sweden. Summary results are shown in Table-2. The authors reported subgroup results for planned cesarean section versus vaginal delivery in their Table-3 (within the article). Regarding sex differences, the authors report "When analyzing hs-cTnT in all infants (irrespective of mode of delivery), male infants had significantly higher values in cord blood than female infants (39[30–51] ng/L vs 28[24–39] ng/L, p = 0.001). However, in samples taken at 2–5 days of age the significant difference disappeared (96[62–157] ng/L vs 85[47–158] ng/L, p = 0.40)."
| Age | n | 99th percentile |
|---|---|---|
| Cord | 105 | 88 |
| 2-5d | 73 | 664 |
- Lam et al (2020)[8]
- Multi-center, outpatient study of 484 healthy outpatient pediatric patients in Toronto, Canada, aged 0 to 19 years. Both hs-cTnT and N-Pro-BNP were studied. Table-3 shows the study's reported age ranges and associated 99th percentile cut-offs for hs-cTnT. Within the article, Fig-1 shows the distribution of troponin versus age with different colors representing male and female values. The authors report finding no difference by sex, but the analysis was probably limited by sample size.
| Age | n | 99th percentile |
|---|---|---|
| 0–<6 m | 64 | 87 |
| 6 m–<1y | 45 | 39 |
| 1–<19 years | 131 | 11 |
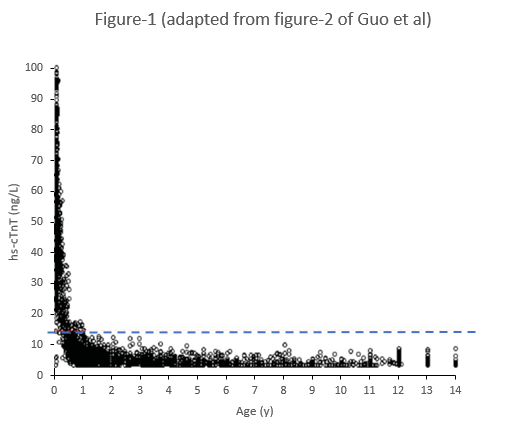
- Guo et al (2021)[9]
- From January 2017 to February 2020, 4,617 questionnaires distributed to children from Chongqing Southeast Hospital (China), pediatricians physically examined respondents to confirm no health issues, 3,463 children included in the study, ages 0-14y, analyzed by sex. Results are presented in Table-4.
- The combination of large sample size and pediatrician physical exams to confirm no underlying health problems makes this undoubtedly the most robust data available at the time of this writing (January 2022).
| Age | n | 99th percentile |
|---|---|---|
| 1d(UCB) | 34 | 60.8 |
| 1d(VB) | 253 | 78.8 |
| 2–28d | 236 | 96.6 |
| 29d–<3m | 178 | 58.6 |
| 3–<6m | 177 | 34.2 |
| 6m–<1y | 377 | 16.2 |
| 1–<3y | 899 | 11.4 |
| 3–<6y (M) | 361 | 8 |
| 3–<6y (F) | 327 | 7.8 |
| 6–14y (M) | 373 | 7.8 |
| 6–14y (F) | 248 | 7.3 |
- Figure-1 is an adaptation of Guo et al's figure illustrating the distribution of hs-cTnT versus age:
Diagnostic Cutoff[s]
99th percentile for a healthy population. Since this is a definition (as discussed above), the term "diagnostic cut-off" does not clearly apply to this cut-off.
Validated Clinical Use[s]
N-Pro-BNP
Biochemistry
Released solely due to myocardial stretch/strain of the left ventricle, which is usually synonymous with heart failure.[REF]
Normal Range[s]
Diagnostic Cutoff[s]
Validated Clinical Use[s]
METHODS
Literature searches were performed for the specific test types available at YKDRH:
- high-sensitivity Troponin T
- N-Terminal pro-BNP
Broader searches, such as for troponin in general are prone to return results for conventional low sensitivity troponin or for troponin-I; data from these are not applicable to interpretation of high-sensitivity troponin-T. Similarly, data regarding B-type natriuretic peptide (BNP) cannot be applied to the test which we have, which is N-Terminal Pro-BNP.
- Troponin-T PubMed Search
- (pediatric*[Title] OR neonat*[Title] OR newborn*[Title] OR infan*[Title] OR child*[Title] or adolescen*[Title]) AND ("high-sensitiv*"[Title] AND "troponin T"[Title]) Search
- with diagnostic outcomes:
- (pediatric*[Title] OR neonat*[Title] OR newborn*[Title] OR infan*[Title] OR child*[Title] or adolescen*[Title]) AND ("high-sensitiv*"[Title] AND "troponin T"[Title]) AND ("receiver operating characteristic"[Title/Abstract] OR "ROC curve"[Title/Abstract] OR AUROC[Title/Abstract] OR specificity[Title/Abstract] OR cut-off[Title/Abstract] OR cutoff[Title/Abstract] OR "Diagnostic accuracy"[Title/Abstract]) Search
- BNP PubMed Search
- (pediatric*[Title] OR neonat*[Title] OR newborn*[Title] OR infant*[Title] OR child*[Title] or adolescen*[Title]) AND ("N-Terminal pro-B-Type Natriuretic Peptide"[Title]) Search
- with diagnostic outcomes:
- (pediatric*[Title] OR neonat*[Title] OR newborn*[Title] OR infant*[Title] OR child*[Title] or adolescen*[Title]) AND ("N-Terminal pro-B-Type Natriuretic Peptide"[Title]) AND ("receiver operating characteristic"[Title/Abstract] OR "ROC curve"[Title/Abstract] OR AUROC[Title/Abstract] OR specificity[Title/Abstract] OR cut-off[Title/Abstract] OR cutoff[Title/Abstract] OR "Diagnostic accuracy"[Title/Abstract]) Search
NOTES
† A percentile cutoff in a healthy population is NOT the same as a diagnostic cutoff; a percentile is an observation detached from clinical implication, whereas diagnostic cut-off has an associated sensitivity and specificity for diagnosing a particular disease/condition.
REFERENCES
- ↑ Jehlicka P, Rajdl D, Sladkova E, Sykorova A, Sykora J. Dynamic changes of high-sensitivity troponin T concentration during infancy: Clinical implications. Physiol Res. 2021 Mar 17;70(1):27-32. doi:10.33549/physiolres.934453. Epub 2021 Jan 14. PMID:33453718. Archived
- ↑ 2.0 2.1 Wang AP, Homme JL, Qureshi MY, Sandoval Y, Jaffe AS. High-Sensitivity Troponin T Testing for Pediatric Patients in the Emergency Department. Pediatr Cardiol. 2021 Nov 17. doi:10.1007/s00246-021-02726-7. Epub ahead of print. PMID:34787696. (available on Sci-Hub)
- ↑ Assandro P, Vidoni M, Starc M, Barbi E. Troponin T should not be considered as a screening test for pediatric myocarditis. Pediatr Emerg Care. 2013 Aug;29(8):955. doi:10.1097/PEC.0b013e31829eca1d. PMID:23925259. (available on Sci-Hub)
- ↑ Ammann P, Pfisterer M, Fehr T, Rickli H. Raised cardiac troponins. BMJ. 2004 May 1;328(7447):1028-9. doi:10.1136/bmj.328.7447.1028. PMID:15117768; PMCID:PMC403831. (available on Sci-Hub)
- ↑ Jehlicka P, Huml M, Rajdl D, Mockova A, Matas M, Dort J, Masopustova A. How to interpret elevated plasmatic level of high-sensitive troponin T in newborns and infants? Physiol Res. 2018 May 4;67(2):191-195. doi:10.33549/physiolres.933704. Epub 2018 Jan 5. PMID:29303610.
- ↑ Bohn MK, Higgins V, Kavsak P, Hoffman B, Adeli K. High-Sensitivity Generation 5 Cardiac Troponin T Sex- and Age-Specific 99th Percentiles in the CALIPER Cohort of Healthy Children and Adolescents. Clin Chem. 2019 Apr;65(4):589-591. doi:10.1373/clinchem.2018.299156. Epub 2019 Feb 8. PMID:30737206. Archived
- ↑ Karlén J, Karlsson M, Eliasson H, Bonamy AE, Halvorsen CP. Cardiac Troponin T in Healthy Full-Term Infants. Pediatr Cardiol. 2019 Dec;40(8):1645-1654. doi:10.1007/s00246-019-02199-9. Epub 2019 Sep 5. PMID:31489446; PMCID:PMC6848050.
- ↑ Lam E, Higgins V, Zhang L, Chan MK, Bohn MK, Trajcevski K, Liu P, Adeli K, Nathan PC. Normative Values of High-Sensitivity Cardiac Troponin T and N-Terminal pro-B-Type Natriuretic Peptide in Children and Adolescents: A Study from the CALIPER Cohort. J Appl Lab Med. 2021 Mar 1;6(2):344-353. doi:10.1093/jalm/jfaa090. PMID:32995884. (available on Sci-Hub)
- ↑ Guo Q, Yang D, Zhou Y, Zhang S, Zhu T, Wang A, Lei M, Yang X. Establishment of the reference interval for high-sensitivity cardiac troponin T in healthy children of Chongqing Nan'an district. Scand J Clin Lab Invest. 2021 Nov;81(7):579-584. doi:10.1080/00365513.2021.1979245. Epub 2021 Sep 28. PMID:34581638. (obtained through the Alaska Medical Library)
- Keywords
- Cardiac Biomarker Troponin BNP Pediatric Neonate Newborn Infant Toddler Child Adolescent
- Author[s]
- Andrew W. Swartz, MD
- Reviewer[s]
- Leslie Herrmann, MD